Crystallization of Mn(II) and Cd(II) Complexes in A Water-Methanol System: Tartrate vs Nicotinamide Ligand Selectivity
DOI:
https://doi.org/10.29303/aca.v5i1.114Keywords:
ligand preference, tartrate, niacinamide, layered solution, metal complexAbstract
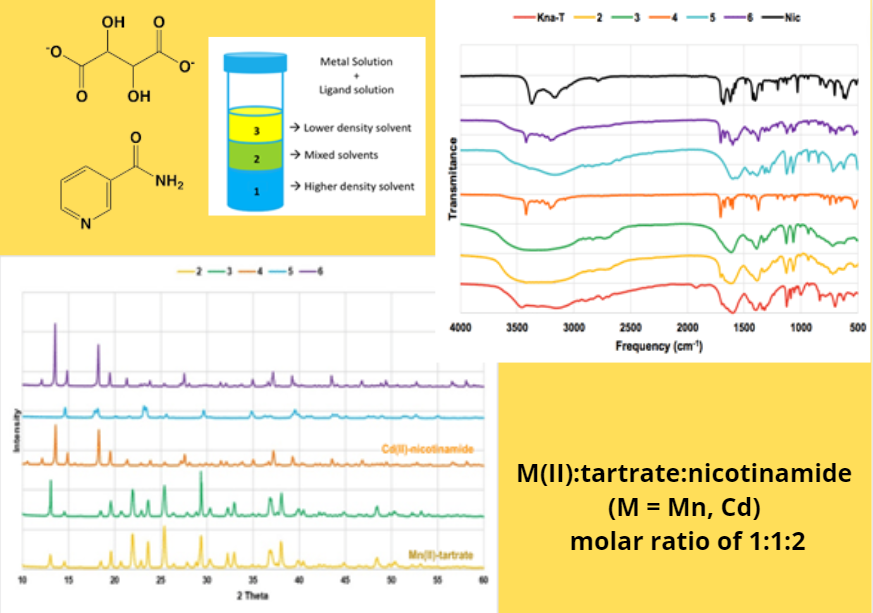
Ligand selectivity of tartrate vs nicotinamide in a water-methanol system has been observed in the crystallization of Mn(II) and Cd(II) complexes. These complexes were crystallized at room temperature by a layered solution technique using a water-methanol mixture solvent in a M(II):tartrate:nicotinamide (M = Mn, Cd) molar ratio of 1:1:2. Complexes of M(II)-nicotinamide and M(II)-tartrate were also prepared for data comparison. Analysis of the crystals by infrared spectroscopy, powder-X-ray diffraction and qualitative anion test showed that in a presence of both tartrate and nicotinamide, the Mn(II) forms neutral Mn(II)-tartrate hydrate complex, whereas the Cd(II) forms ionic Cd(II)-nicotinamide chloride complex. In the case of Mn(II) complex, tartrate tend to coordinate as ligand than the nicotinamide, although molar ratio of nicotinamide was doubled than that of tartrate ligand. In contrast, the neutral nicotinamide ligand is more predominant to coordinate in the Cd(II) complex than the anionic tartrate. The tartrate-nicotinamide ligand selectivity in the crystallization of Mn(II) and Cd(II) complexes is likely due to the use of tartrate salt as precursor and the choice of solvent mixture. In addition, powder-XRD analysis confirms that there was no indication of M(II)-tartrate and M(II)-nicotinamide that co-crystallized together at the same time by both metal ions.
Downloads
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Yuniar Ponco Prananto, Rafi Dwiasis Wibisono, Sasti Gona Fadhilah, Rachmat Triandi Tjahjanto, Darjito, Firsta Luthfiani Salsabila

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Authors who publish with ACA: Acta Chimica Asiana agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. This license allows authors to use all articles, data sets, graphics, and appendices in data mining applications, search engines, web sites, blogs, and other platforms by providing an appropriate reference. The journal allows the author(s) to hold the copyright without restrictions and will retain publishing rights without restrictions.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in ACA: Acta Chimica Asiana.
- Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access).





 Indonesian Chemical Society, Chapter Nusa Tenggara. Jalan Majapahit 62 Mataram, University of Mataram, 83125, Indonesia
Indonesian Chemical Society, Chapter Nusa Tenggara. Jalan Majapahit 62 Mataram, University of Mataram, 83125, Indonesia
